On June 28th we visited a small finger of Mýrdalsjökull (“mire valley glacier”) called Solheimajokull. Our goal was to stream accurate elevation data from our ambiance platforms in order to approximate the extent of the glacial finger. If our data comes out as we hope, we can combine it with existing datasets to approximate the volume of Solheimajokull. We were able to walk reasonably far up the glacier as well as to each edge, so we are hopeful that the volume calculation will be possible.

On Wednesday June 29th our group bifurcated for the day to take care of two separate tasks. One group returned to Solheimajokull to take soil samples in the glacial forefield and the other stayed at the airbnb to work on platform reliability at data visualization.
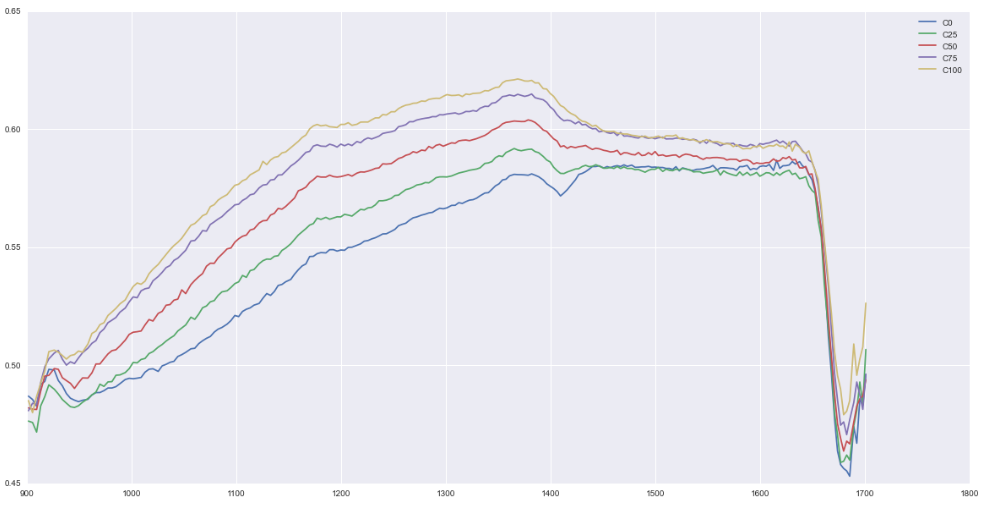
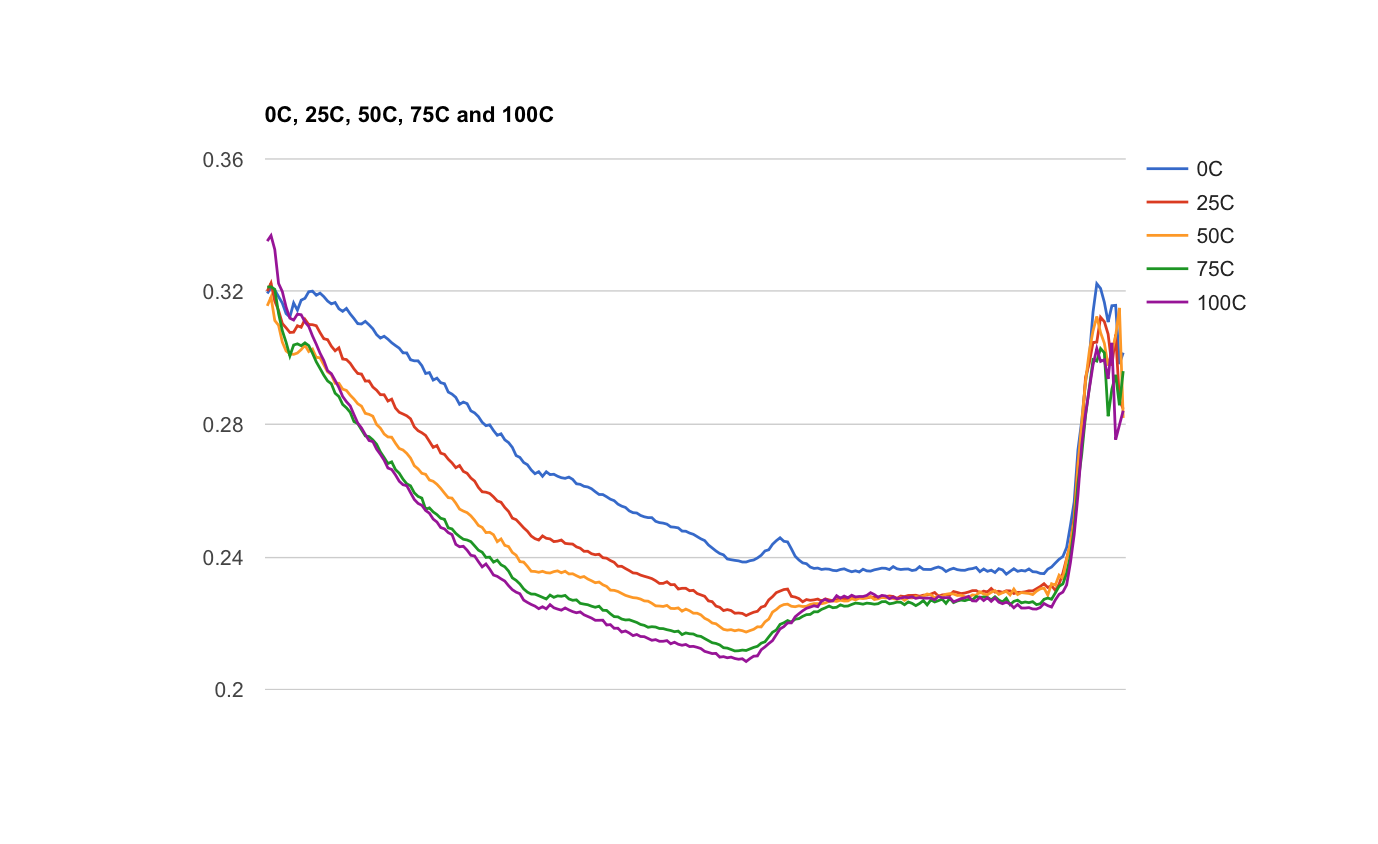
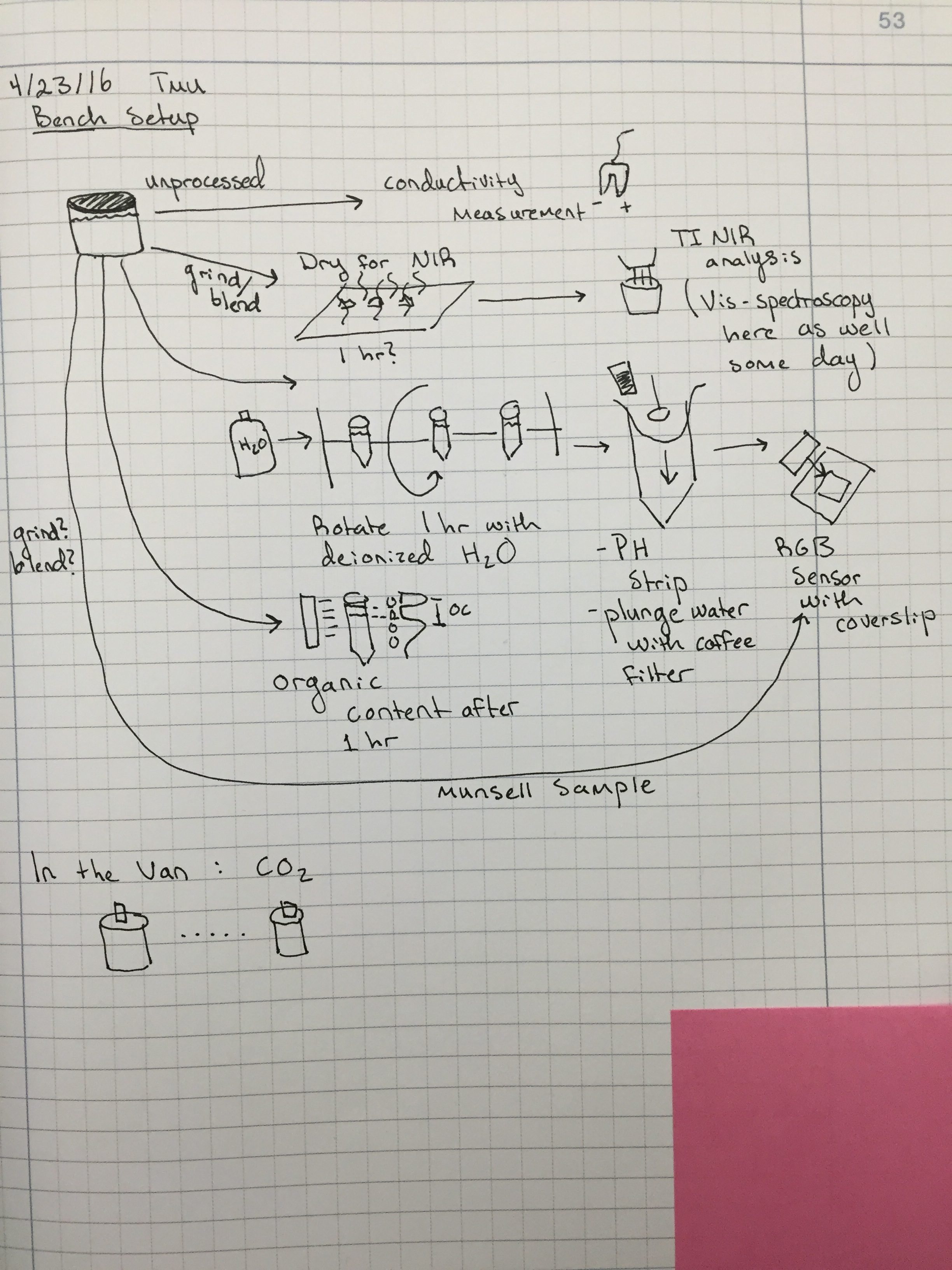
The forefield represents a chronosequence in which the soil has developed as the glacier recedes. Our goal is to observe how soil composition varies with age. While sampling, we observed changes in the smell and texture of the soil as well as increasing plant growth as we got farther from the glacier. In the evening we separated out rocks from the soil and carried out NPK (nitrogen, potassium, and phosphorous) testing on the first few samples. In other words, we sat around our kitchen table and shook dirt in tubes for a few hours. In the next few days we will test the remainder of the soil samples as well as characterize the carbon concentration in the soil using NIR spectroscopy.

On the 30th we traveled east, making two notable stops. The first was at Grafarkirkja, a river at which the tephra layers created by volcanic eruptions are visible. We took samples from the six topmost layers. Our goal is to identify the eruption of Laki based on the unique chemical signature of its ash. The ash produced in the eruption of Laki had unusually high levels of fluorine. With this information we should be able to distinguish Laki’s tephra layer from the other layers.
Our second stop was at the picturesque ‘Glacier Lagoon’, which is fed by Iceland’s largest glacier Vatnajökull. Large chunks of ice can be seen floating in the water. Charlie observed that there are fewer ice chunks than in 2014 and that the heavy recession of Vatnajökull is apparent across the lagoon.

Today we arrived at Skálanes and have begun setting up our gear for carrying out soil analysis. At Skálanes we will conduct surveys of the nearby archeological site using our UAV and LiDAR platform. We are also interested in surveying local bird and fox populations using the UAV. All of the geocoded images and data we record will contribute to our goal of orienting our results using QGIS. We will also take soil samples from the gardens here to determine the its composition.